Pico : Can Airway Obstruction Measured By Spirometry Help Diagnose Asthma In Adults With Episodic/chronic Suggestive Symptoms
Recommendation
-
The TF recommends performing spirometry to detect airway obstruction as part of the diagnostic work-up of adults aged 18years with suspected asthma
Remarks
-
An FEV1/FVC < LLN or < 75%, higher than the commonly utilised 70% threshold, should be considered supportive of an asthma diagnosis and should prompt further testing
-
A normal spirometry does not exclude asthma
Background
Spirometry is a non-invasive physiological test, performed since the 19th Century, that measures the volume and flow of air during inhalation and exhalation. A standardised procedure for performing spirometry has been published by the ERS and the ATS . The ratio of the forced expiratory volume in the first second to the forced vital capacity is an index reflecting airway obstruction. The TF assessed the FEV1/FVC ratio to determine whether it could help in the diagnosis of asthma.
Review of the evidence
Our literature search identified 11 potentially relevant studies of which four were suitable to be included , all performed in secondary care that assessed the accuracy of the FEV1/FVC ratio to predict the probability of asthma ascertained by either BdR of 12% and 200mL or 15% reversibility, methacholine BHR , or 20% PEF variability over a two-week period .
National Asthma Guidelines Updated
New Approaches for Monitoring Asthma Control, Expanded Recommendations for Children.
The National Asthma Education and Prevention Program today issued the first comprehensive update in a decade of clinical guidelines for the diagnosis and management of asthma. The guidelines emphasize the importance of asthma control and introduce new approaches for monitoring asthma. Updated recommendations for managing asthma include an expanded section on childhood asthma with an additional age group, new guidance on medications, new recommendations on patient education in settings beyond the physician’s office and new advice for controlling environmental factors that can cause asthma symptoms.
Coordinated by the National Heart, Lung, and Blood Institute of the National Institutes of Health, NAEPP convenes an expert panel when there is sufficient science to warrant a rigorous, systematic review of the published medical literature to ensure that the asthma guidelines reflect the latest scientific advances.
Asthma is one of the most common health problems in the United States – and it can significantly affect patients’ lives – at school, at work, at play, and at home, said NHLBI Director Elizabeth G. Nabel, M.D. It is essential that asthma patients benefit from the best available scientific evidence, and these guidelines bring such evidence to clinical practice.
Key features and changes to these four components of asthma care include:
Resources:
NIHTurning Discovery Into Health®
Metotrexate Antifungal Agents Macrolides
There are low evidences in the efficacy of metotrexate in children with asthma. A meta-analysis investigating the steroid-sparing effect of oral metotrexate in asthmatics showed a small benefit despite a high recurrence of adverse effect .
Antifungal agents are not recommended in asthmatic patients without allergic broncopulmonary aspergillosis . Vicencio and coworkers recently documented that a high percentage of children with refractory asthma had fungal sensitization correlated to the severity of their disease. Based to a case report of a positive response to itraconazole treatment of a child with severe asthma, the Authors suggest that antifungal therapy may represent a potential successful treatment in some patients with evidence of fungal sensitization, after eliminating molds in the environment .
Recommended Reading: Is Asthma Chronic Lung Disease
National Asthma Guidelines Get First Update In 13 Years
The guidance, which includes 19 recommendations, was published in Journal of Allergy and Clinical Immunology. It represents the first updates to federal comprehensive asthma management and treatment guidelines in more than a decade.
The last national guidance on asthma care was published 13 years ago, and since then weve made substantial progress in understanding how to treat asthma in children and adults, Michelle M. Cloutier, MD, professor emerita, University of Connecticut School of Medicine, and chair of the National Asthma Education Prevention Program Coordinating Committee Expert Panel Working Group, said in a statement. In addition to asthma management varying by age group and disease severity, the preferences and values that individuals with asthma place on different therapies must be considered. The new guidelines reflect some of these new approaches.
The 6 areas of guidance with updates are:
- Using inhaled corticosteroids when needed for recurrent wheezing or persistent asthma
- Using long-acting antimuscarinic antagonists with inhaled corticosteroids for long-term asthma
The recommendations are based on systemic reviews by the Agency for Healthcare Research and Quality from research published before October 2018. In her foreword to the guidelines, Cloutier noted that there is an absence of many strong recommendations, which are those recommendations that clinicians should adhere to almost all the time as standard of care.
Reference
Guidelines For The Diagnosis And Management Of Asthma 2007
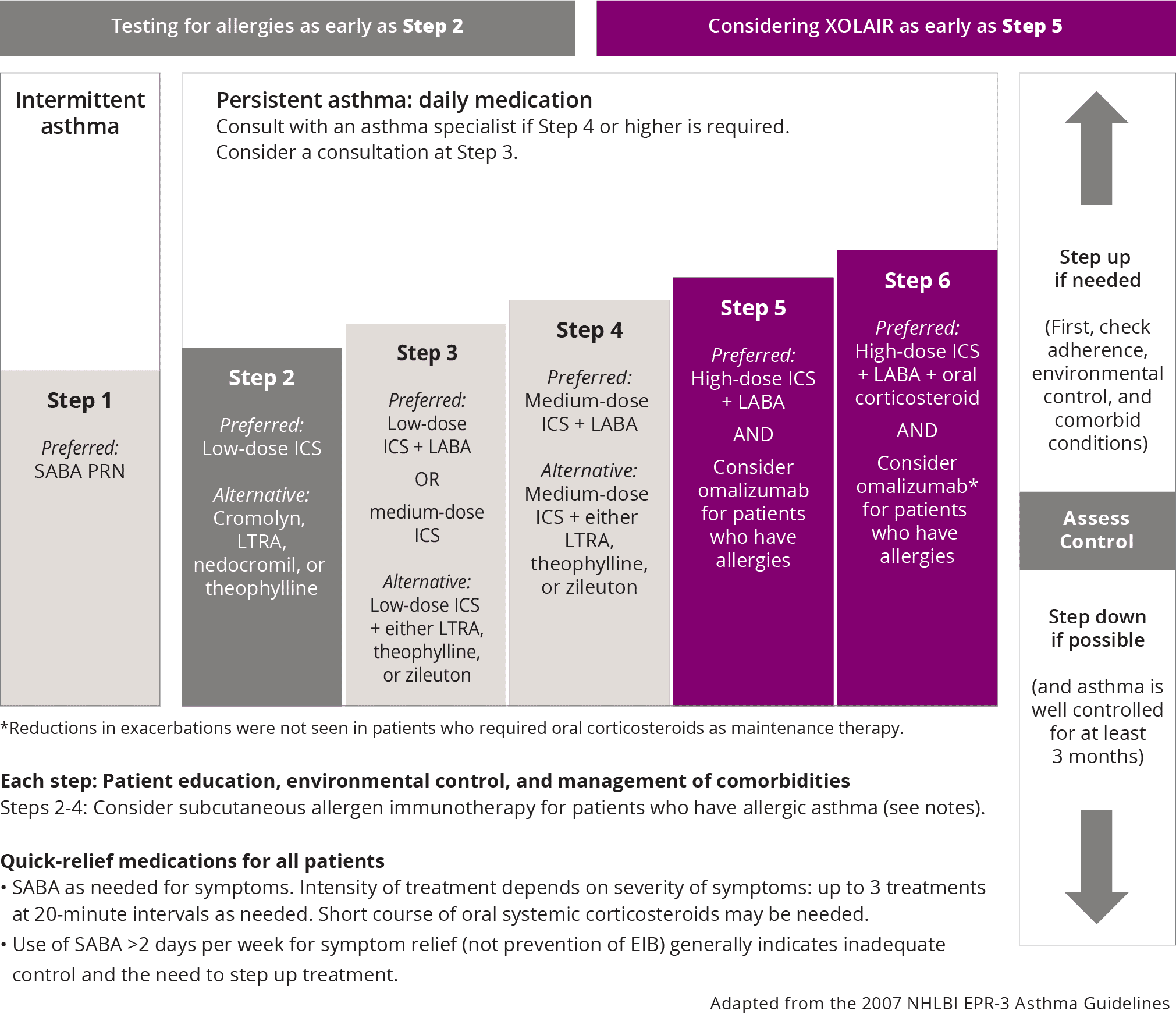
The EPR 3 Guidelines on Asthma was developed by an expert panel commissioned by the National Asthma Education and Prevention Program Coordinating Committee , coordinated by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Using the 1997 EPR 2 guidelines and the 2004 update of EPR 2 as the framework, the expert panel organized the literature review and final guidelines report around four essential components of asthma care, namely: assessment and monitoring, patient education, control of factors contributing to asthma severity, and pharmacologic treatment. Subtopics were developed for each of these four broad categories.
Version History:
- EPR-2 Update on Selected Topic in 2002
- EPR-2 published in 1997
- EPR-1 published in 1991
The EPR 3 Guidelines on Asthma was developed by an expert panel commissioned by the National Asthma Education and Prevention Program Coordinating Committee , coordinated by the National Heart, Lung, and Blood Institute of the National Institutes of Health. Using the 1997 EPR 2 guidelines and the 2004 update of EPR 2 as the framework, the expert panel organized the literature review and final guidelines report around four essential components of asthma care, namely: assessment and monitoring, patient education, control of factors contributing to asthma severity, and pharmacologic treatment. Subtopics were developed for each of these four broad categories.
Version History:
Recommended Reading: Does A Steamy Shower Help Asthma
Categories Of Asthma Medications And Step
The pharmacological options for treatment of asthma include, according to their use, reliever medications, which are drugs that allow relief of symptoms within few minutes, during worsening asthma or exacerbations, also used for prevention of exercise-induced bronchoconstriction controller medications, that are used for maintenance treatment: they control symptoms and reduce airway inflammation and future risks of exacerbations add-on therapies, proposed for patients with severe persistent asthma symptoms and exacerbations, despite treatment with high dose controller medications.
The most used drug classes of asthma medications are beta-2 adrenergic agonists, corticosteroids, and leukotriene modifiers, usually montelukast.
A stepwise approach for pharmacotherapy management in asthmatic patients has been proposed .1). Treatment starts at the step most appropriate to the initial severity of asthma. If control is not achieved within 3 months, stepping-up should be tried, after reconsidering adherence to therapy, environment factors, and associated co-morbidities otherwise, treatment step down may be attempted once good asthma control and the patient’s lowest effective level of treatment has been found and maintained for about 3 months. Each recommendation has been assessed for adults adolescents and children in all guidelines .
Financial And Other Disclosures
Development of the resource document and the guidelines report was funded by the NHLBI, NIH. Expert Panel members completed financial disclosure forms, and the Expert Panel members disclosed relevant financial interests to each other prior to their discussions. Expert Panel members participated as volunteers and were compensated only for travel expenses related to the Expert Panel meetings. Financial disclosure information covering the 3 year period during which the guidelines were developed is provided for each Panel member below.
Deceased
Read Also: Can You Develop Asthma When Pregnant
Limitations Of 2020 Focused Updates
The expert panel kept its focus on asthma. The immunotherapy recommendations apply only to their role in asthma and not other elements of allergy care. The updates do not address biologics because the evidence for biologics was limited when topics were selected. Cost of care was acknowledged, but it was out of scope to address the complex nature of health care payment and insurance coverage. The updates did note the potential impact on disparities for recommended tests and treatments within each topic area.
These Focused Updates are an important resource for navigating the increasingly complex care options for asthma. The expert panel invites feedback from family physicians on these recommendations and other areas where significant clinical questions persist .
